 |
Alex GUMOVSKY
Ph.D., Dr.Sci.
Schmalhausen Institute of Zoology
15 Bogdan Khmelnitsky Street
Kiev-30
01030
UKRAINE
Tel.: +38044 234-9333
E-mail:
gumovsky@izan.kiev.ua
entedon@gmail.com
|
SCIENTIFIC INTERESTS
 My research focuses
on the systematics, phylogeny and biogeography of the Chalcidoidea (Hymenoptera).
Chalcidoid wasps is one of the largest groups of insects: there are about as 100,000
described species, but many are still undescribed and regional faunas are also
poorly known. Some of chalcids are specialist, but many are also generalist parasitoids;
lots are used in successful biological control programs against insect pests. My research focuses
on the systematics, phylogeny and biogeography of the Chalcidoidea (Hymenoptera).
Chalcidoid wasps is one of the largest groups of insects: there are about as 100,000
described species, but many are still undescribed and regional faunas are also
poorly known. Some of chalcids are specialist, but many are also generalist parasitoids;
lots are used in successful biological control programs against insect pests.
One area of my research is the family Eulophidae, the
most numerous group of Chalcidoidea (ca. 4470 described species). The taxonomy
of this group is relatively poorly understood. Eulophids attack wide variety of
insect, and occasionally mite or spider hosts. Their larvae act as koino- or idiobionts,
gregarious or solitary, ecto- or endoparasitoids; they attack eggs, larvae or
pupae of their hosts. Some species are phytophagous (chiefly gall-formers on eucalypts).
 A second area of research
is the family Tetracampidae. This is a small family with about 50 described
species classified in 15 genera. The extant groups of the family are represented
by egg parasitoids of Hymenoptera and Coleoptera (Tetracampinae), larval parasitoids
of Diptera (Tetracampinae, Platynocheilinae), and phytophagous species (Mongolocampinae).
Unlike Eulophidae, the Tetracampidae look rather arteficial than natural group
in its current concept. A second area of research
is the family Tetracampidae. This is a small family with about 50 described
species classified in 15 genera. The extant groups of the family are represented
by egg parasitoids of Hymenoptera and Coleoptera (Tetracampinae), larval parasitoids
of Diptera (Tetracampinae, Platynocheilinae), and phytophagous species (Mongolocampinae).
Unlike Eulophidae, the Tetracampidae look rather arteficial than natural group
in its current concept.
Another area of my interests is fossil Chalcidoidea. Chalcids
are relatively poorly known from the fossils. The amber inclusions are probably
the only source for such fossil data because of minute size and weak body sclerotization
of these insects.
 There are many reasons
for the comparatively modest progress in this area: poor condition of the amber
samples, minute diagnostic characters not visible in amber specimens, poor representation
of the chalcids in amber fossils (in comparison with other groups of insects),
to list some. There are many reasons
for the comparatively modest progress in this area: poor condition of the amber
samples, minute diagnostic characters not visible in amber specimens, poor representation
of the chalcids in amber fossils (in comparison with other groups of insects),
to list some.
The records of Eulophidae from ambers are rare.
Boucek & Askew (1968) briefly mentioned Entedon sp. from Dominican
amber, however, without exact identification of the amber specimen.
Then Gumovsky (2001) recorded two eulophids also from Dominican
amber (Chrysonotomyia dominicana and Achrysocharoides
sp.).
Some amber insect inclusions are classified in Tetracampidae:
namely this family is among the most represented ones in fossil resins. However,
clarification of the correct family affiliation of fossil tetracampids requires
additional studies. The placement of fossil groups in Tetracampidae was likely
motivated by the conventional concept of this family as intermediate between Pteromalidae
and Eulophidae, and thus serving as a “dumping ground” for the species with problematic
status.
 My
current research in this field focuses on a survey of chalcid inclusions
in the Canadian and Taymyr (Cretaceous, 80-90 million years ago), Baltic
(Late Eocene, about 40 million years ago) and Dominican (Miocene, about
20 million years ago) ambers. Gumovsky et al. (2018) conducted a survey
of the chalcidoid inclusions in Taimyr amber (84-100 Ma). As a result,
and 11 new species, were described in Baeomorpha. Also, Baeomorphinae
Yoshimoto, 1975, based on Baeomorpha Brues, 1937, is transferred
from Tetracampidae Foerster, 1856 and recognized as a junior synonym
of Rotoitidae Boucek and Noyes, 1987 (Hymenoptera: Chalcidoidea). One
enigmatic rotoitid inclusion, which differs from Baeomorpha species
in the possession of very short stigmal vein, is described as Taimyromorpha
pusilla Gumovsky. Inclusions containing specimens identified as
Baeomorpha and Taimyromorpha are found in amber from Taimyr
and Canada that originated from Laurasia, not Gondwana. The newly described
fossils indicate the southern hemisphere distribution of extant Rotoitidae
is relictual with the pattern observed being formed at least in part
by extinction events, though distributions of the only two extant rotoitid
genera, Rotoita (New Zealand) and Chiloe (small area in
the southern Chile) may have been more extensive in the past. Also,
the Canadian amber genera Distylopus (Distylopinae) and Bouceklytus
(Bouceklytinae) are excluded from Tetracampidae and regarded as Chalcidoidea
incertae sedis. My
current research in this field focuses on a survey of chalcid inclusions
in the Canadian and Taymyr (Cretaceous, 80-90 million years ago), Baltic
(Late Eocene, about 40 million years ago) and Dominican (Miocene, about
20 million years ago) ambers. Gumovsky et al. (2018) conducted a survey
of the chalcidoid inclusions in Taimyr amber (84-100 Ma). As a result,
and 11 new species, were described in Baeomorpha. Also, Baeomorphinae
Yoshimoto, 1975, based on Baeomorpha Brues, 1937, is transferred
from Tetracampidae Foerster, 1856 and recognized as a junior synonym
of Rotoitidae Boucek and Noyes, 1987 (Hymenoptera: Chalcidoidea). One
enigmatic rotoitid inclusion, which differs from Baeomorpha species
in the possession of very short stigmal vein, is described as Taimyromorpha
pusilla Gumovsky. Inclusions containing specimens identified as
Baeomorpha and Taimyromorpha are found in amber from Taimyr
and Canada that originated from Laurasia, not Gondwana. The newly described
fossils indicate the southern hemisphere distribution of extant Rotoitidae
is relictual with the pattern observed being formed at least in part
by extinction events, though distributions of the only two extant rotoitid
genera, Rotoita (New Zealand) and Chiloe (small area in
the southern Chile) may have been more extensive in the past. Also,
the Canadian amber genera Distylopus (Distylopinae) and Bouceklytus
(Bouceklytinae) are excluded from Tetracampidae and regarded as Chalcidoidea
incertae sedis.
Other my research interests include functional morphology
of attachment and grooming structures in Hymenoptera. One of the structures is
the pretarsus, that serves as the main attachment device. The pretarsus of Chalcidoidea
is characterized by a distal elastic widening of the planta that spreads over
the arcus, by a pair of folding plates at the dorsal side of the arolium (the
dorsal plates), and by the absence of auxiliary sclerites. The surface of the
fully spread arolium of chalcids has a spongiform structure. The peculiarities
of the inverting/everting biomechanics of the pretarsus of chalcids involve: (1)
interactions between the elastic part of the planta, the dorsal plates and the
manubrium; (2) the functioning of the elastic part of the planta and the arcus
together as a single unit.
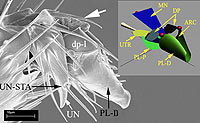 A
manubrium with a distinct proximal row of three setae characterizes almost all
Eulophidae, Aphelinidae and Signiphoridae (‘eulophid lineage’) and Tetracampidae,
whereas a row of two setae characterizes Mymaridae, Rotoitidae and Trichogrammatidae.
Other studied families (Pteromalidae, Eurytomidae, Torymidae, Ormyridae, Eupelmidae,
Encyrtidae, Perilampidae), which represent a ‘pteromalid lineage ’, are characterized
mostly by five setae in a proximal row, which could represent a synapomorphy for
these groups, or a symplesiomorphy in Chalcidoidea, depending on rooting. However,
these characters may be correlated with differences in body size that characterize
the different lineages rather than being phylogenetically important. Other characters
that may be phylogenetically informative are: (1) shape of the manubrium (spindle-like
in Mymaridae, Rotoitidae, Trichogrammatidae and the ‘eulophid lineage’, but mostly
bottle-like in representatives of the ‘pteromalid lineage’); (2) pubescence of
the proximal part of the planta (sparse, thick setae in Rotoitidae, Trichogrammatidae
and the ‘eulophid lineage’, but dense, slender setae in representatives of the
‘pteromalid lineage’). A
manubrium with a distinct proximal row of three setae characterizes almost all
Eulophidae, Aphelinidae and Signiphoridae (‘eulophid lineage’) and Tetracampidae,
whereas a row of two setae characterizes Mymaridae, Rotoitidae and Trichogrammatidae.
Other studied families (Pteromalidae, Eurytomidae, Torymidae, Ormyridae, Eupelmidae,
Encyrtidae, Perilampidae), which represent a ‘pteromalid lineage ’, are characterized
mostly by five setae in a proximal row, which could represent a synapomorphy for
these groups, or a symplesiomorphy in Chalcidoidea, depending on rooting. However,
these characters may be correlated with differences in body size that characterize
the different lineages rather than being phylogenetically important. Other characters
that may be phylogenetically informative are: (1) shape of the manubrium (spindle-like
in Mymaridae, Rotoitidae, Trichogrammatidae and the ‘eulophid lineage’, but mostly
bottle-like in representatives of the ‘pteromalid lineage’); (2) pubescence of
the proximal part of the planta (sparse, thick setae in Rotoitidae, Trichogrammatidae
and the ‘eulophid lineage’, but dense, slender setae in representatives of the
‘pteromalid lineage’).
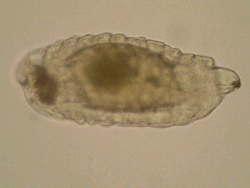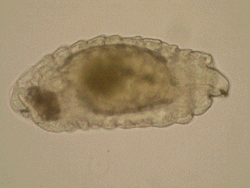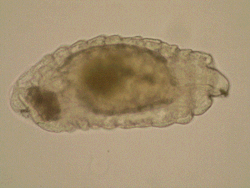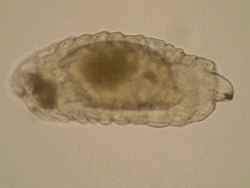 One
more area of my research interests include morphology of immature
stages of Chalcidoidea and all aspects of biology and evolution
of egg-larval parasitism in Chalcidoidea, which are rather fragmentary
up-to-date. Most discussions on larval morphology and biology
of chalcid wasps concern ectoparasitoids, whereas endoparasitoid
larvae traditionally attract less attention, mainly due to the
difficulties with their preparation and identification. One
more area of my research interests include morphology of immature
stages of Chalcidoidea and all aspects of biology and evolution
of egg-larval parasitism in Chalcidoidea, which are rather fragmentary
up-to-date. Most discussions on larval morphology and biology
of chalcid wasps concern ectoparasitoids, whereas endoparasitoid
larvae traditionally attract less attention, mainly due to the
difficulties with their preparation and identification.
The development of solitary
and gregarious eulophid endoparasitoids (Entedon, in
particular) differs in immature morphology and physiology. Generally,
the first-instar larvae of solitary species have a beak-shaped
head with sharp mandibles and get involved in siblicide (killing
of rivals). The first-instar larvae of gregarious endoparasitoids
are characterized by shorter mandibles and are tolerant to each
other (Harvey et al. 2012), although the morphology of the larval
gregarious parasitoids of the genus Entedon has not been
described so far. Gumovsky & Ramadan (2011) described a unique
biology of Afrotropical species Entedon erythrinae, a
gregarious egg-larval endoparasitoid of the bruchid beetle Specularius
impressithorax.
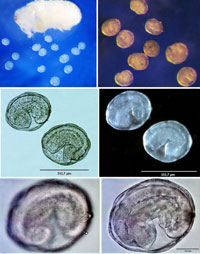 The
early development of E. erythrinae differs from all other
species of the genus and is peculiar among known life histories
of chalcidoid wasps. Unlike other non-polyembryonic chalcidoids,
the early development of the parasitoid egg is not associated
with organogenesis, but simply results in multiplication of proliferating
cells. The cells differentiate into the external layer (formed
by the larger elongate cells) and the inner cell mass (formed
by the smaller relatively rounded cells). The external layer gives
rise to an extraembryonic membrane or serosa (so called ‘soccer
ball’ chamber), while the internal cell aggregation develops further
into the larva. This larva shares all the morphological characters
with the second instar of other Entedon species, for
which immature stages are known. Therefore, the first (aggressive
and responsible for siblicide) instar larval stage is omitted,
what may happen to be an adaptation to gregariousness (Gumovsky
& Ramadan 2011). However, more data on other gregarious endoparasitoids
are required to verify whether such embryonization is an ultimate
or exceptional adaptation. The
early development of E. erythrinae differs from all other
species of the genus and is peculiar among known life histories
of chalcidoid wasps. Unlike other non-polyembryonic chalcidoids,
the early development of the parasitoid egg is not associated
with organogenesis, but simply results in multiplication of proliferating
cells. The cells differentiate into the external layer (formed
by the larger elongate cells) and the inner cell mass (formed
by the smaller relatively rounded cells). The external layer gives
rise to an extraembryonic membrane or serosa (so called ‘soccer
ball’ chamber), while the internal cell aggregation develops further
into the larva. This larva shares all the morphological characters
with the second instar of other Entedon species, for
which immature stages are known. Therefore, the first (aggressive
and responsible for siblicide) instar larval stage is omitted,
what may happen to be an adaptation to gregariousness (Gumovsky
& Ramadan 2011). However, more data on other gregarious endoparasitoids
are required to verify whether such embryonization is an ultimate
or exceptional adaptation.
The origin and alteration
of the egg-larval and larval parasitism within the same genus
of parasitoids may shed light on the evolution of aggressiveness
and tolerance in animal world as a whole (with possible implications
for humans also: e.g., Gomez et al. 2016).
All these studies incorporate morphological, biological or
molecular information into analyses that are used to produce hypotheses of
phylogenetic relationships and the evolution of behavioral patterns.
The first signal of monophyly of Eulophidae
was obtained by Campbell et al. (2000) based on 28S D2 rDNA gene
of chalcidoids. Also, the genus Elasmus, the only genus of the
former family Elasmidae, appeared a derived taxon within Eulophidae.
Then Gauthier et al. (2000) used the same gene on broader sampling
to clarify the phylogeny of Eulophidae. These authors supported
the idea that Elasmidae are derived eulophids, the subfamilies
Eulophinae and Tetrastichinae are closely related and provided
some new characters to support monophyly of some groups of Eulophidae.
Gumovsky (2002) also used the same gene for the subfamily Entedoninae
and discussed distribution of some morphological characters in
Entedoninae.
The families Eulophidae, Tetracampidae, Trichogrammatidae
and also Aphelinidae are sometimes considered as the “eulophid
lineage” (Gibson et al., 1997). This group is characterized by
reduced number of antennal (generally 4-7, but occasionally more)
and tarsal (3-4) segments. However, there are many overlaps in
distribution of these characters as within the “eulophid lineage”,
as with outgroups. Gladun & Gumovsky (2006) demonstrated that
the representatives of the “eulophid lineage” share similar morphology
of pretarsus. Gokhman (2004, 2009) and then Gokhman & Gumovsky
(2009) suggested that Eulophidae belong to the so-called "low-numbered"
chalcidoid families with the modal haploid number of chromosomes
n=6 (but with occasional modifications to 5 or 7), whereas n=9-12
in many other families. However, such karyoptype occurs also outside
Eulophidae and also the number of chromosomes reduced independently
in various groups of Chalcidoidea (Gokhman & Gumovsky, 2009).
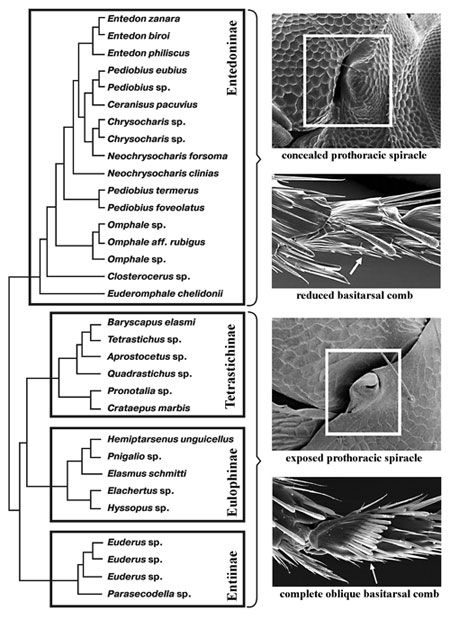 Our
research (Gumovsky, 2011) based on the combined analysis of nuclear
(28S D2 rDNA) and two mitochondrial (cytochrome oxidase subunit
I, COI, and cytochrome b, Cyt b) sequences demonstrated that there
is no signal of close relationships between the families Eulophidae,
Tetracampidae and Trichogrammatidae, but at least Eulophidae and
Trichogrammatidae are supported as monophyletic. Also, the Eulophidae-lineage
appeared to consist of four internal lineages corresponding to
the recognized subfamilies: Eulophinae, Tetrastichinae, Entedoninae
and Entiinae (= Euderinae), what was also supported by morphological
characters (including putative synapomorphies). Largely similar
pattern was reported by other students foe Eulophidae (Burks et
al., 2011) and Chalcidoidea as a whole (Munro et al., 2011; Heraty
et al., 2013). Our
research (Gumovsky, 2011) based on the combined analysis of nuclear
(28S D2 rDNA) and two mitochondrial (cytochrome oxidase subunit
I, COI, and cytochrome b, Cyt b) sequences demonstrated that there
is no signal of close relationships between the families Eulophidae,
Tetracampidae and Trichogrammatidae, but at least Eulophidae and
Trichogrammatidae are supported as monophyletic. Also, the Eulophidae-lineage
appeared to consist of four internal lineages corresponding to
the recognized subfamilies: Eulophinae, Tetrastichinae, Entedoninae
and Entiinae (= Euderinae), what was also supported by morphological
characters (including putative synapomorphies). Largely similar
pattern was reported by other students foe Eulophidae (Burks et
al., 2011) and Chalcidoidea as a whole (Munro et al., 2011; Heraty
et al., 2013).
The most ancient records of fossil Chalcidoidea
are dated as Cretaceous (78-115 Mya) and represented by the ‘basal’
group (e.g. Mymarommatidae and Mymaridae and the unspecified ‘aphelinid–trichogrammatid’
representatives: Heraty et al., 2013). The rest of the groups
are common in Eocene deposits (ca. 40 Mya), and so the main diversification
is presumed to have taken place in the Late Cretaceous or soon
after. The phylogenetic analyses conducted so far (Burks et al.,
2011; Munro et al., 2011; Heraty et al., 2013) suggest that the
common ancestor of Eulophidae, Tetracampidae and Trichogrammatidae
had branched off yet in Gondwana (ca. 120–60 Mya). In particular,
the enigmatic genus Trisecodes (unplaced within Eulophidae) had
diversified when the modern Africa and South America were not
yet split (Gumovsky, 2014).
Research on functional morphology of Hymenoptera has been used
for phylogenetic interpretation. Gibson (1985) and Heraty et al. (1997) discussed
the phylogenetic implications of some pro- and mesothoracic structures. Basibuyuk
et al. also inferred phylogenetic information from the morphology of the antennal
cleaner (1995), hamuli (1997) and grooming behaviours (1999). Basibuyuk et al.
(2000a) also discussed peculiarities of the structure and location of the sensilla
on the manubrium in Hymenoptera in light of established hypotheses of relationships
within Hymenoptera.
Gladun & Gumovsky (2006) demonstrated that character-state
distribution of features of the pretarsus among studied taxa support some established
groups or proposed associations of Chalcidoidea.
The evolution of host associations and peculiarities of host
shifts during natural history of the group, are fascinating areas of research.
Such studies are also of paramount importance to biocontrol programs: this demonstrates
the utility and impact of systematics to almost every area of science.
Fellowships and research visits
2016 - visiting researcher,
IZIKO, South African National Museum, Cape Town, South Africa.
2014, 2015, 2016 - visiting
researcher, University of Witwatersrand, Johannesburg, South Africa.
2013 - University of Kisangani
(UNIKIS) - Centre de Surveillance de la Biodiversite, Democratic
Republic of Congo: collaborative research.
http://www.africamuseum.be/museum/research/general/backfromafrica/back-gumovsky
2012-2013 - Post-doctoral
fellow, the Museum of Central Africa (Tervuren) (MRAC-RMCA), BELSPO-Marie
Curie supported fellowship.
http://www.africamuseum.be/museum/research/general/backfromafrica/back-gumovsky
2010-2011 - Wageningen University
and The Netherlands Institute of Ecology (NIOO-KNAW)
2009 - Bishop Museum, Honolulu, Hawaii, USA:
a study of collection material.
2009 - University of Hawaii at Manoa, Department of
Entomology, Honolulu, Hawaii, USA: a workshop with students and a study of collection
material.
2009 - Hawaii Deapartment of Agriculture, Honolulu, USA,
a collaborative project on invasive bruchid beetle pests and their parasitoids.
2009 - ICIPE, Nairobi, Kenya: a study of collection material
2008 - Mugla University, Mugla, Turkey: workshop with
students, a study of collection material, collaborative research and field trips.
2008 - University of Dakar, Senegal: a study of collection
material, collaborative research and field trips.
2007 - Mustafa Kemal University, Antakya, Turkey: a study
of collection material, collaborative research and field trips
2006 - Paleontological Institute of Russian Academy of
Sciences (Moscow, Russia): a study of fossil chalcidoids.
2005 - Entomology Research Museum, University of California
(Riverside, USA): a study of collection material.
2005 - The Natural History Museum (London, UK): a study
of collection material, molecular studies on eulophid wasps.
2004 - Naturkunde Museum (Stuttgart, Germany): a study
of chalcidoid inclusions in fossil resins.
2004 - Zoologische Staatssammlung (Munchen, Germany):
a study of collection material.
2004 - Max-Planck Institute for Metal Research (Stuttgart,
Germany): a study on functional morphology of chalcids.
2003 - European Station of CABI Bioscience (Delemont,
Switzerland): biocontrol project management.
2000 - CIRAD-Amis (Montpellier, France): a study of collection
material.
2000 - Museum National d'Histoire Naturelle (Paris, France):
a study of collection material.
2000 - Zoological Muzeum, University of Copenhagen (Denmark):
a study of collection material.
2000 - The Natural History Museum (London, UK): a study
of collection material.
2000 - Imperial College at Silwood Park (Ascot, UK): molecular
studies on eulophid wasps.
1998 - Zoological Institute of Russian Academy of Sciences
(St.-Petersburg, Russia): a study of collection material.
1996 - Regional Environmental Center for Eastern and Central
Europe (Budapest, Hungary): nature conservation project management.
Scientific grants
2014 - SYNTHESYS Project:
the research project was conducted in The Natural History Museum
(London).
2014 - The award of the State
Fund of Fundamental Research (Ukraine), grant F50/029 for young
holders of Dr. Sci. (Habilitation) degree: Parasitoid guilds of
alien pests as a model for studying of the adaptive capacities
of native and alien parasitoids.
2012-2013 - BELSPO-Marie Curie
supported fellowship.
2011 - The award of the State
Fund of Fundamental Research (Ukraine), grant F35/002 for young
holders of Dr. Sci. (Habilitation) degree: Phylogenetic and faunistic
research on Eulophidae (Hymenoptera).
2010-2011 - Wageningen University
‘Production Ecology and Resource Centre’ (PE&RC, Netherlands)
Visiting Scientist Scholarship.
2011 - SFFR (The State Fund of Fundamental
Research (Ukraine)), grant F 35/002.
2007- 2009 - Alexander von Humboldt Foundation,
research fellowship award.
2006 - TUBITAK / NASU Academic Exchange grant.
2005 - SYNTHESYS Project (GB–TAF–535).
2004 - DAAD (German Academic Exchange Service, grant
No. 322 – A/04/15867).
2001 - SFFR (The State Fund of Fundamental Research (Ukraine))
grant 05.07/00078.
2000 - European Commission research grant (under COBICE-programme).
1999 - Royal Society / NATO Grant (NATO/ 99A/bll).
Awards
2014 – I.I. Schmalhausen Award of the
National Academy of Sciences of Ukraine (together with A. Radchenko
and E.E. Perkovsky).
2002-2004 - President of Ukraine Scholarship
for young scientists.
1997, 1998 - George Soros Foundation (International Foundation
Vidrodgenia) grants.
1996 - The National Academy of sciences of Ukraine award
for young scientists.
1997 - Travel grant of the International Association of
Academies of Sciences.
1995 - Taras Shevchenko’s award of Kiev National University.
1995 - Special student award of Kiev National University.
Participation in international projects on phylogeny and taxonomy
2016-2018 - NSF An integrative
approach to understanding the evolution and systematics of Chalcidoidea:
A recent megaradiation of Hymenoptera (superviser Prof. J. Heraty,
University of California, Riverside). Contribution to the project:
phylogeny of Eulophidae, Tetracampidae.
2005-2009 - Tree of Life Project: Phylogeny
of Chalcidoidea (superviser Prof. J. Heraty, University of California,
Riverside). Contribution to the project: phylogeny of Eulophidae,
Tetracampidae.
2006 - A project “Identifications of taxa of Entedoninae
(Hym.: Eulophidae) from Turkey by using molecular DNA markers” supported by TUBITAK
/ NASU Academic Joint Research grant, in collaboration with Mustafa Kemal University,
Antakya, Turkey. Contribution to the project: taxonomic and phylogenetic studies.
Nature conservation grants / Projects
1997 - Biodiversity Support Program (U.S.A.I.D. - funded
consortium of World Wildlife Fund, The Nature Conservancy, and World Resources
Institute): “Studying on and conservation of beneficial insects (Hymenoptera mainly)
of the National Park “Podol’s’ki Tovtry” (South-Western Ukraine) [Project Director].
1994 - ISAR, a Clearinghouse on Grassroots Cooperation
in Eurasia: “Biodiversity survey in the nature reserve "Lesniki" (Kiev
vicinity)” [Project Director].
Participation in biocontrol projects
2009 - A project on biology of an invasive bruchid beetle
pest Specularius impressithorax and its hymenopterous parasitoids (with Hawaii
Deapartment of Agriculture, Honolulu, USA). Project contribution: studies biology,
development and taxonomy of the beetle and its parasitoids.
2003-2005 - A project on approaches to control invasive
moth pest Cameraria ohridella (curated by Schmalhausen Institute of Zoology, Kiev,
Ukraine). Project contribution: studies on biology and taxonomy of the parasitoids
of the moth.
2002-2004 - Institutional Partnership project ‘‘Emphasising
Classical and Conservation Biological Control in Research and Teaching’’ (7 IP
65648). Project contribution: preparation of manuals on biocontrol; lectures and
studies on biology of biocontrol agents (beetles and parasitoids).
2001-2002 - A project on approaches to control invasive
moth pest Acrocercops brongniardella (curated by Schmalhausen Institute of Zoology,
Kiev, Ukraine). Project contribution: studies on biology and taxonomy of the parasitoids
of the moth.
Fieldwork
Ukraine (1995-2016)
England, France (2000)
Hungary (2001, 2010)
Turkey (2007, 2008)
Senegal (2008)
The Democratic Republic of Congo (2013, 2014,
2015)
Uganda (2009, 2013, 2014, 2015, 2016)
Tanzania (2008)
Kenya (2009, 2013)
Hawaii (2009)
Zambia (2011)
Botswana (2016)
Namibia (2011, 2014-2016)
South Africa (2014-2016)
Services
Anonymous referee in “Zoosystematica Rossica", "Entomotropica",
"Zootaxa", “Journal of Natural history”.
Languages
Ukrainian (native), Russian (second native), English (fluent),
German (functional).
SELECTED PUBLICATIONS
(in reverse order, PDFs available upon request)
Monograph
Gumovsky, A. (2023). Revision of Xiphentedon Risbec,
1957 and Colpixys Waterston, 1916 (Hymenoptera: Eulophidae), with descriptions
of new species from the Afrotropics. European Journal of Taxonomy, 905(1):
1–83. DOI: https://doi.org/10.5852/ejt.2023.905.2325
Book
Gumovsky, A.V. 2012. The family Eulophidae
(Hymenoptera: Chalcidoidea): systematics, morphology and life histories.
Kiev, Naukova Dumka. 216 pp. (in Ukrainian)
Chapters in Monographs
In: Heraty, J. & Woolley, J.B. (Eds.).
Chalcidoidea of the World (2025). CABI Publishing. 840 pp. ISBN (print):
9781800623521.
• Chapter 4. Natural History of Chalcidoidea. By: John M. Heraty,
Petr Jansta, Paul Hanson, Jean-Yves Rasplus, Alex Gumovsky, John S.
Noyes, James B. Woolley, Gary A.P. Gibson. pp. 41–68.
• Chapter 11. Baeomorphidae. By: John T. Huber, John Heraty,
Alex Gumovsky, Lars Krogmann. pp. 222–228.
• Chapter 27. Eulophidae. By: Roger A. Burks, Alex Gumovsky,
Christer Hansson, Michael W. Gates, Tiffany Domer, Ryan K. Perry. pp.
357–375.
• Chapter 58. Tetracampidae. By: Alex Gumovsky, Roger A. Burks,
Serguei V. Triapitsyn. pp. 621–630.
Articles in journals with peer review
Cruaud, A., Rasplus, J.-Y., Zhang, J., Burks, R., Delvare,
G., Fusu, L., Gumovsky, A., Huber, J.T., Jansta, P., Mitroiu, M.-D.,
Noyes, J.S., van Noort, S., Baker, A., Bohmova, J., Baur, H., Blaimer,
B.B., Brady, S.G., Bubenikova, K., Chartois, M., Copeland, R.S., Dale-Skey
Papilloud, N., Dal Molin, A., Dominguez, C., Gebiola, M., Guerrieri,
E., Kresslein, R.L., Krogmann, L., Lemmon, E., Murray, E.A., Nidelet,
S., Nieves-Aldrey, J.L., Perry, R.K., Peters, R.S., Polaszek, A., Saune,
L., Torrens, J., Triapitsyn, S., Tselikh, E.V., Yoder, M., Lemmon, A.R.,
Woolley, J.B., Heraty, J.M. 2024. The Chalcidoidea bush of life:
evolutionary history of a massive radiation of minute wasps. Cladistics,
40(1), 34–63. https://doi.org/10.1111/cla.12561
Gumovsky, A. 2023. A new genus of Chalcidoidea
(Hymenoptera) from Chile with challenging taxonomic position. Zootaxa,
5254(1), 133–141. https://doi.org/10.11646/zootaxa.5254.1.8
Gumovsky, A., van Noort, S.V. 2022. Afronympha,
a new genus of Entedoninae (Hymenoptera: Eulophidae) from the rainforests
of Africa. Zootaxa, 5104(2), 242–250.
Tymochko, L.I., Cuaranhua, C., Gumovsky, A.V. 2021.
Habitat distribution of diapriid parasitoid wasps (Hymenoptera, Diaprioidea)
in eastern Mozambique. International Journal of Tropical Insect Science,
41, 3187–3201.
Gumovsky, A. 2021. Review of the species of
Pediobius (Hymenoptera, Eulophidae) having extreme dorsal setation
and description of a new species from East Africa. Zootaxa, 4999(5):
423–438.
Gumovsky, A., Bazhenova, T., van Noort, S. 2020.
First record of the genus Setelacher Bouček (Hymenoptera:
Eulophidae: Eulophinae) from the Afrotropical region, with description
of a new species. Journal of Natural History, 54(9-12): 611-620. [abstract]
Gumovsky, A.V. 2020. Biological notes on Ceranisus
menes (Hymenoptera: Eulophidae: Entedoninae), a biological control
agent of thrips, in Ukraine. Ukrainska Entomofaunistyka, 11(3): 5-10.
[abstract]
Rasplus, J.-Y., Blaimer, B.B., Brady, S.G., Burks,
R. G., Delvare, G., Fisher, N., Gates, M., Gauthier, N., Gumovsky, A.V.,
Hansson, C., Heraty, J.M., Fusu, L., Nidelet, S., Pereira, R.A.S., Sauné,
L., Ubaidillah, R., Cruaud, A. 2020. A first phylogenomic hypothesis
for Eulophidae (Hymenoptera, Chalcidoidea). Journal of Natural History,
54(9-12): 597-609. [abstract]
Cruaud, A., Nidelet, S., Arnal, P., Weber, A., Fusu,
L., Gumovsky, A., Huber, J., Polaszek, A., Rasplus, J.-Y. 2019.
Optimised DNA extraction and library preparation for minute arthropods:
application to target enrichment in chalcid wasps used for biocontrol.
Molecular Ecology Resources 19(3): 702-710. [abstract]
Woelke, J. B., Fursov, V. N., Gumovsky, A. V., de Rijk,
M., Estrada, C., Verbaarschot, C., Huigens, M. E., Fatouros, N. E. 2019.
Description and biology of two new egg parasitoid species (Hymenoptera:
Trichogrammatidae) reared from eggs of Heliconiini butterflies (Lepidoptera:
Nymphalidae: Heliconiinae) in Panama. Journal of Natural History 53:
11-12, 639-657; DOI: 10.1080/00222933.2019.1606360. [abstract]
Gumovsky, A., Perkovsky, E., Rasnitsyn, A. 2018.
Laurasian ancestors and "Gondwanan" descendants of Rotoitidae
(Hymenoptera: Chalcidoidea): What a review of Late Cretaceous Baeomorpha
revealed". Cretaceous Research 84: 286–322. [abstract]
Gumovsky, A. 2018. New enigmatic species of
the genus Pediobius (Hymenoptera, Eulophidae) from Afrotropics, with
notes on related genera. Zootaxa 4438(2): 201–236.
[abstract]
Gumovsky A. 2018. A new genus of Tetracampidae
(Hymenoptera) from South Africa. European Journal of Taxonomy 447: 1–13.
[abstract]
Jaloszynski, P., Gumovsky, A. 2017. Four species
of Entedon Dalman (Hymenoptera: Chalcidoidea: Eulophidae) new in the
fauna of Poland. Wiadomosci Entomologiczne 36(1): 54–58. [abstract]
Gumovsky, A. 2016. Review of Afrotropical
species of Goetheana Girault (Hymenoptera: Eulophidae), with description
of a new species. Zootaxa, 4147 (5): 551–563. [abstract]
Gumovsky, A. 2016. A review
of genera and described Afrotropical species of Tetracampinae
(Hymenoptera: Tetracampidae), with description of a new genus
from East Central Africa. Zootaxa, 4111 (4): 393–420. [abstract]
Matushkina N.A., Riasanov S.A., Shyriaieva
D.A., Gorobchishin V.A., Gumovsky A.V. 2016.
Tritrophic association on dandelion in a European capital city:
pilot quantitative study on the herbivore weevil Glocianus punctiger
and its parasitoid wasp Entedon costalis. Urban Ecosystems, 19
(3): 1119–1130. [abstract]
Gumovsky, A., De Little, D., Rothman, S.,
Jaques, L., Mayorga, S.E.I. 2015. Re-description
and first host and biology records of Entedon magnificus (Girault
& Dodd) (Hymenoptera, Eulophidae), a natural enemy of Gonipterus
weevils (Coleoptera, Curculionidae), a pest of Eucalyptus trees.
Zootaxa 3957 (5): 577–584. [abstract]
De Prins, J., Gumovsky, A., De Coninck, E.
2015. Discovery of a new species of Caloptilia
(Lepidoptera: Gracillariidae) from east and central Africa with
its suggested associated host (Gentianales: Rubiaceae) and natural
enemies (Hymenoptera: Eulophidae). Zootaxa 3957 (4): 383–407.
[abstract]
Gumovsky, A. 2014. A new
species of Trisecodes from the Afrotropics (Hymenoptera, Eulophidae).
Zootaxa. 3852: 553-61.[abstract]
Poelman, E.H., Gols, R., Gumovsky, A.V., Cortesero,
A.-M., Dicke, M., Harvey, J.A. 2014. Food plant
and herbivore host species affect the outcome of intrinsic competition
among parasitoid larvae. Ecological Entomology, 39: 693–702. [abstract]
Tamesse, J.L., Soufo, L., Tchanatame, E.C.,
Dzokou, V.J., Gumovsky, A., De Coninck, E. 2014.
Description of Psyllaephagus blastopsyllae sp.n. (Encyrtidae),
new species, endoparasitoid of Blastopsylla occidentalis Taylor
(Psyllidae, Spondyliaspidinae) in Cameroon. Journal of Biodiversity
and Environmental Sciences, 5(2), 227-236. 
Heraty, J.M., Burks, R.A., Cruaud, A., Gibson,
G.A.P., Liljeblad, J., Munro, J., Rasplus, J.-Y., Delvare, G.,
Jansta, P., Gumovsky, A., Huber, J., Woolley, J.B., Krogmann,
L., Heydon, S., Polaszek, A., Schmidt, S., Darling, D.C., Gates,
M.E., Mottern, J., Murray, E., DalMolin, A., Triapitsyn, S., Baur,
H., Pinto, J.D., van Noort, S., George, J., Yoder, M.A. 2013.
A phylogenetic analysis of the megadiverse Chalcidoidea (Hymenoptera).
Cladistics, 29: 466–542. [abstract]
Gumovsky, A.V., Simutnik, S.A., Prokhorov,
A.V. 2013. Life-history review of Oobius zahaikevitshi
Trjapitzin, 1963 (Hymenoptera: Encyrtidae), an egg parasitoid
of jewel beetles (Coleoptera: Buprestidae). Russian Entomological
Journal, 22(3): 181–188. 
Bolsheva, N.L., Gokhman, V.E., Muravenko,
O.V., Gumovsky, A.V., Zelenin, A.V. 2012. Comparative
cytogenetic study on two species of the genus Entedon Dalman,
1820 (Hymenoptera, Eulophidae) using DNA-binding fluorochromes
and molecular and immunofluorescent markers. Comparative Cytogenetics,
6(1): 79–92. [abstract]
Harvey, J.A., Gumovsky, A., Gols, R. 2012.
Effect of host-cocoon mass on adult size in the secondary hyperparasitoid
wasp, Pteromalus semotus (Hymenoptera: Pteromalidae). Insect Science,
19(3): 383–390. [abstract]
Gokhman, V.E., Gumovsky, A.V. 2012.
New data on chromosomes of Chalcidoidea (Hymenoptera). Zoological
Journal, 91(8): 975-979. [abstract]
Gumovsky, A.V. 2011. Molecular
data support the existence of four main lineages in the phylogeny
of Eulophidae (Hymenoptera). Russian Entomological Journal, 20(3):
273-286. 
Gumovsky, A., Ramadan, M. 2011.
Biology, immature and adult morphology, and molecular characterisation
of a new species of the genus Entedon (Hymenoptera: Eulophidae)
associated with the invasive pest Specularius impressithorax (Coleoptera:
Chrysomelidae, Bruchinae) on Erythrina plants. Bulletin of Entomological
Research, 101: 715-739. [abstract]
Gumovsky A. V. 2010. A record
of Sinoxylon anale Lesne in Ukraine with notes on false powder-post
beetles (Coleoptera: Bostrichidae) and their chalcidoid parasitoids
(Hymenoptera). Ukrainska Entomofaunistyka, 2: 1-8. 
Gokhman, V.E, Gumovsky, A.V. 2009. Main trends
of karyotype evolution in the superfamily Chalcidoidea (Hymenoptera).
Comparative Cytogenetics, 3(1): 63-69.
Gumovsky, A. 2008. Parasitism of Entedon costalis (Hymenoptera:
Eulophidae) in Glocianus punctiger (Coleoptera: Curculionidae): an example of
intentional discovery of the parasitoid-host association. Zootaxa, 1964: 40-68

Gumovsky A.V. 2007. A taxonomic revision, biology and
morphology of immature stages of the Entedon sparetus species group (Hymenoptera:
Eulophidae), egg-larval endoparasitoids of weevils (Coleoptera: Curculionidae).
Bulletin of Entomological Research, 97: 139–166. 
Gumovsky, A. 2007. Taxonomic notes on genera allied
to Pleurotroppopsis (Hymenoptera: Eulophidae, Entedoninae) with description of
a new genus from the Afrotropical region. Zootaxa, 1415: 1-16.  (abstract)
(abstract)
Gumovsky, A., Rusina, L., Firman, L. 2007. Bionomics
and morphological and molecular characterization of Elasmus schmitti and Baryscapus
elasmi (Hymenoptera: Chalcidoidea, Eulophidae), parasitoids associated with a
paper wasp, Polistes dominulus (Vespoidea, Vespidae). Entomological Science, 10:
21-34. 
Gladun, D., Gumovsky, A. 2006. The pretarsus in Chalcidoidea
(Hymenoptera Parasitica): functional morphology and possible phylogenetic implications.
Zoologica Scripta, 35(6): 607–626. 
Gumovsky, A., 2006. The biology and morphology of Entedon
sylvestris (Hymenoptera: Eulophidae), a larval endoparasitoid of Ceutorhynchus
sisymbrii (Coleoptera: Curculionidae). Journal of Hymenoptera Research 15(2):
232-250.  (abstract)
(abstract)
Gumovsky, A., Polaszek, A., Murphy, S., Rabbi, M.F., Zhu, C.-D.
2006. Closterocerus oryzamyntor (Hymenoptera: Eulophidae: Entedoninae),
a larval parasitoid of the rice hispa Dicladispa armigera in Bangladesh (Coleoptera:
Chrysomelidae: Hispinae). Zootaxa, 1241: 51–59. 
Gumovsky, A., Boucek, Z., Delvare, G. 2006. New genera
and species of Afrotropical Entedoninae (Hymenoptera, Eulophidae). Zoologische
Mededelingen, Leiden, 80-1(4): 73-85. 
Gumovsky, A.V., Perkovsky, E.E. 2005. Taxonomic notes
on Tetracampidae (Hymenoptera: Chalcidoidea) with description of a new fossil
species of Dipricocampe from Rovno amber. Entomological Problems, 35(2): 123-130.

Gumovsky, A.V., Boucek, Z. 2005. A new genus of Entedoninae
(Hymenoptera, Eulophidae) from Malaysia, associated with ant nests. Entomological
Problems, 35(1): 39-42.  (abstract)
(abstract)
Gumovsky, A.V. 2005. Observations on foraging and mating
behaviour of Euderomphale chelidonii (Hymenoptera: Eulophidae). Acta Societatis
Zoologicae Bohemicae, 69: 101-108.
Gumovsky A. V. 2004. Studies on the DNA sequence data
in systematics: general issues and a model protocol. The Kharkov Entomological
Society Gazette, 11(1-2): 213-236 [Гумовський О.В. 2003 (2004). Дослідження структури
ДНК в систематиці: загальні положення та модельний протокол. Известия Харьковского
энтомологического общества, 11(1-2): 213-236] (in Ukrainian, with English summary
and supplement)
Gumovsky, A.V. 2003. Review of the Pediobius alcaeus species
group (Insecta: Hymenoptera: Eulophidae: Entedoninae) with taxonomic notes on
related genera and the description of a new species from Japan. Species Diversity,
8: 275-292.
Gumovsky, A.V., Boyadzhiev, P. 2003. Review of the Bulgarian
Entedon Dalman, 1820 (Hymenoptera: Eulophidae, Entedoninae). Acta Zoologica Bulgarica,
55(3): 3-32.  (abstract)
(abstract)
Gumovsky, A.V. 2003. Review and taxonomic notes on the
genus Derostenus (Hymenoptera, Eulophidae). Vestnik zoologii, 37(6): 3-14.
Gumovsky, A., Boucek, Z. 2003. A new genus of Entedoninae
(Hymenoptera: Eulophidae) from Brazil. Neotropical Entomology, 32(3): 443-447.

Gumovsky, A.V. 2003. The status of Kokandia Yefremova
et Kriskovich (Hymenoptera: Eulophidae), with taxonomic notes on some related
genera. Zoologische Mededelingen, Leiden, 77(12): 245-251. 
Gumovsky, A.V. 2003. New peculiar entedonine genus (Hymenoptera:
Chalcidoidea: Eulophidae) from Western Australia. Australian Journal of Entomology,
42(1): 79-83. 
Gumovsky, A. 2002. Monophyly and preliminary phylogeny
of Entedoninae (Hymenoptera, Chalcidoidea, Eulophidae): 28S D2 rDNA considerations
and morphological support / in Parasitic Wasps: Evolution, Systematics, Biodiversity
and Biological Control (G. Melika and Cs. Thuroczy, eds.), Agroinfrom, Budapest,
Hungary. P.193-219. 
Gumovsky, A., Ubaidillah, R. 2002. Revision of the genus
Parzaommomyia Girault (Hymenoptera: Eulophidae), with taxonomic notes on related
genera. Zoologische Mededelingen, Leiden, 76 (9): 93-132. 
Gumovsky, A. 2001. Taxonomic notes on the entedonine
genera Rhynchentedon and Pediobomyia (Hymenoptera: Chalcidoidea: Eulophidae) with
the description of a new species. Zoologische Mededelingen, Leiden, 75 (14): 229–238.

Gumovsky A.V. 2001. The status of some genera allied
to Chrysonotomyia Ashmead and Closterocerus Westwood (Hymenoptera, Eulophidae,
Entedoninae), with description of a new species from Dominican Amber. Phegea,
29 (4): 125-141. 
Gumovsky A.V. 2001. Review of the genus Paracrias (Hymenoptera,
Eulophidae). Vestnik zoologii, 35 (5): 9-26.
|

 My research focuses
on the systematics, phylogeny and biogeography of the Chalcidoidea (Hymenoptera).
Chalcidoid wasps is one of the largest groups of insects: there are about as 100,000
described species, but many are still undescribed and regional faunas are also
poorly known. Some of chalcids are specialist, but many are also generalist parasitoids;
lots are used in successful biological control programs against insect pests.
My research focuses
on the systematics, phylogeny and biogeography of the Chalcidoidea (Hymenoptera).
Chalcidoid wasps is one of the largest groups of insects: there are about as 100,000
described species, but many are still undescribed and regional faunas are also
poorly known. Some of chalcids are specialist, but many are also generalist parasitoids;
lots are used in successful biological control programs against insect pests. A second area of research
is the family Tetracampidae. This is a small family with about 50 described
species classified in 15 genera. The extant groups of the family are represented
by egg parasitoids of Hymenoptera and Coleoptera (Tetracampinae), larval parasitoids
of Diptera (Tetracampinae, Platynocheilinae), and phytophagous species (Mongolocampinae).
Unlike Eulophidae, the Tetracampidae look rather arteficial than natural group
in its current concept.
A second area of research
is the family Tetracampidae. This is a small family with about 50 described
species classified in 15 genera. The extant groups of the family are represented
by egg parasitoids of Hymenoptera and Coleoptera (Tetracampinae), larval parasitoids
of Diptera (Tetracampinae, Platynocheilinae), and phytophagous species (Mongolocampinae).
Unlike Eulophidae, the Tetracampidae look rather arteficial than natural group
in its current concept. There are many reasons
for the comparatively modest progress in this area: poor condition of the amber
samples, minute diagnostic characters not visible in amber specimens, poor representation
of the chalcids in amber fossils (in comparison with other groups of insects),
to list some.
There are many reasons
for the comparatively modest progress in this area: poor condition of the amber
samples, minute diagnostic characters not visible in amber specimens, poor representation
of the chalcids in amber fossils (in comparison with other groups of insects),
to list some. My
current research in this field focuses on a survey of chalcid inclusions
in the Canadian and Taymyr (Cretaceous, 80-90 million years ago), Baltic
(Late Eocene, about 40 million years ago) and Dominican (Miocene, about
20 million years ago) ambers. Gumovsky et al. (2018) conducted a survey
of the chalcidoid inclusions in Taimyr amber (84-100 Ma). As a result,
and 11 new species, were described in Baeomorpha. Also, Baeomorphinae
Yoshimoto, 1975, based on Baeomorpha Brues, 1937, is transferred
from Tetracampidae Foerster, 1856 and recognized as a junior synonym
of Rotoitidae Boucek and Noyes, 1987 (Hymenoptera: Chalcidoidea). One
enigmatic rotoitid inclusion, which differs from Baeomorpha species
in the possession of very short stigmal vein, is described as Taimyromorpha
pusilla Gumovsky. Inclusions containing specimens identified as
Baeomorpha and Taimyromorpha are found in amber from Taimyr
and Canada that originated from Laurasia, not Gondwana. The newly described
fossils indicate the southern hemisphere distribution of extant Rotoitidae
is relictual with the pattern observed being formed at least in part
by extinction events, though distributions of the only two extant rotoitid
genera, Rotoita (New Zealand) and Chiloe (small area in
the southern Chile) may have been more extensive in the past. Also,
the Canadian amber genera Distylopus (Distylopinae) and Bouceklytus
(Bouceklytinae) are excluded from Tetracampidae and regarded as Chalcidoidea
incertae sedis.
My
current research in this field focuses on a survey of chalcid inclusions
in the Canadian and Taymyr (Cretaceous, 80-90 million years ago), Baltic
(Late Eocene, about 40 million years ago) and Dominican (Miocene, about
20 million years ago) ambers. Gumovsky et al. (2018) conducted a survey
of the chalcidoid inclusions in Taimyr amber (84-100 Ma). As a result,
and 11 new species, were described in Baeomorpha. Also, Baeomorphinae
Yoshimoto, 1975, based on Baeomorpha Brues, 1937, is transferred
from Tetracampidae Foerster, 1856 and recognized as a junior synonym
of Rotoitidae Boucek and Noyes, 1987 (Hymenoptera: Chalcidoidea). One
enigmatic rotoitid inclusion, which differs from Baeomorpha species
in the possession of very short stigmal vein, is described as Taimyromorpha
pusilla Gumovsky. Inclusions containing specimens identified as
Baeomorpha and Taimyromorpha are found in amber from Taimyr
and Canada that originated from Laurasia, not Gondwana. The newly described
fossils indicate the southern hemisphere distribution of extant Rotoitidae
is relictual with the pattern observed being formed at least in part
by extinction events, though distributions of the only two extant rotoitid
genera, Rotoita (New Zealand) and Chiloe (small area in
the southern Chile) may have been more extensive in the past. Also,
the Canadian amber genera Distylopus (Distylopinae) and Bouceklytus
(Bouceklytinae) are excluded from Tetracampidae and regarded as Chalcidoidea
incertae sedis. A
manubrium with a distinct proximal row of three setae characterizes almost all
Eulophidae, Aphelinidae and Signiphoridae (‘eulophid lineage’) and Tetracampidae,
whereas a row of two setae characterizes Mymaridae, Rotoitidae and Trichogrammatidae.
Other studied families (Pteromalidae, Eurytomidae, Torymidae, Ormyridae, Eupelmidae,
Encyrtidae, Perilampidae), which represent a ‘pteromalid lineage ’, are characterized
mostly by five setae in a proximal row, which could represent a synapomorphy for
these groups, or a symplesiomorphy in Chalcidoidea, depending on rooting. However,
these characters may be correlated with differences in body size that characterize
the different lineages rather than being phylogenetically important. Other characters
that may be phylogenetically informative are: (1) shape of the manubrium (spindle-like
in Mymaridae, Rotoitidae, Trichogrammatidae and the ‘eulophid lineage’, but mostly
bottle-like in representatives of the ‘pteromalid lineage’); (2) pubescence of
the proximal part of the planta (sparse, thick setae in Rotoitidae, Trichogrammatidae
and the ‘eulophid lineage’, but dense, slender setae in representatives of the
‘pteromalid lineage’).
A
manubrium with a distinct proximal row of three setae characterizes almost all
Eulophidae, Aphelinidae and Signiphoridae (‘eulophid lineage’) and Tetracampidae,
whereas a row of two setae characterizes Mymaridae, Rotoitidae and Trichogrammatidae.
Other studied families (Pteromalidae, Eurytomidae, Torymidae, Ormyridae, Eupelmidae,
Encyrtidae, Perilampidae), which represent a ‘pteromalid lineage ’, are characterized
mostly by five setae in a proximal row, which could represent a synapomorphy for
these groups, or a symplesiomorphy in Chalcidoidea, depending on rooting. However,
these characters may be correlated with differences in body size that characterize
the different lineages rather than being phylogenetically important. Other characters
that may be phylogenetically informative are: (1) shape of the manubrium (spindle-like
in Mymaridae, Rotoitidae, Trichogrammatidae and the ‘eulophid lineage’, but mostly
bottle-like in representatives of the ‘pteromalid lineage’); (2) pubescence of
the proximal part of the planta (sparse, thick setae in Rotoitidae, Trichogrammatidae
and the ‘eulophid lineage’, but dense, slender setae in representatives of the
‘pteromalid lineage’).  One
more area of my research interests include morphology of immature
stages of Chalcidoidea and all aspects of biology and evolution
of egg-larval parasitism in Chalcidoidea, which are rather fragmentary
up-to-date. Most discussions on larval morphology and biology
of chalcid wasps concern ectoparasitoids, whereas endoparasitoid
larvae traditionally attract less attention, mainly due to the
difficulties with their preparation and identification.
One
more area of my research interests include morphology of immature
stages of Chalcidoidea and all aspects of biology and evolution
of egg-larval parasitism in Chalcidoidea, which are rather fragmentary
up-to-date. Most discussions on larval morphology and biology
of chalcid wasps concern ectoparasitoids, whereas endoparasitoid
larvae traditionally attract less attention, mainly due to the
difficulties with their preparation and identification.  The
early development of E. erythrinae differs from all other
species of the genus and is peculiar among known life histories
of chalcidoid wasps. Unlike other non-polyembryonic chalcidoids,
the early development of the parasitoid egg is not associated
with organogenesis, but simply results in multiplication of proliferating
cells. The cells differentiate into the external layer (formed
by the larger elongate cells) and the inner cell mass (formed
by the smaller relatively rounded cells). The external layer gives
rise to an extraembryonic membrane or serosa (so called ‘soccer
ball’ chamber), while the internal cell aggregation develops further
into the larva. This larva shares all the morphological characters
with the second instar of other Entedon species, for
which immature stages are known. Therefore, the first (aggressive
and responsible for siblicide) instar larval stage is omitted,
what may happen to be an adaptation to gregariousness (Gumovsky
& Ramadan 2011). However, more data on other gregarious endoparasitoids
are required to verify whether such embryonization is an ultimate
or exceptional adaptation.
The
early development of E. erythrinae differs from all other
species of the genus and is peculiar among known life histories
of chalcidoid wasps. Unlike other non-polyembryonic chalcidoids,
the early development of the parasitoid egg is not associated
with organogenesis, but simply results in multiplication of proliferating
cells. The cells differentiate into the external layer (formed
by the larger elongate cells) and the inner cell mass (formed
by the smaller relatively rounded cells). The external layer gives
rise to an extraembryonic membrane or serosa (so called ‘soccer
ball’ chamber), while the internal cell aggregation develops further
into the larva. This larva shares all the morphological characters
with the second instar of other Entedon species, for
which immature stages are known. Therefore, the first (aggressive
and responsible for siblicide) instar larval stage is omitted,
what may happen to be an adaptation to gregariousness (Gumovsky
& Ramadan 2011). However, more data on other gregarious endoparasitoids
are required to verify whether such embryonization is an ultimate
or exceptional adaptation. Our
research (Gumovsky, 2011) based on the combined analysis of nuclear
(28S D2 rDNA) and two mitochondrial (cytochrome oxidase subunit
I, COI, and cytochrome b, Cyt b) sequences demonstrated that there
is no signal of close relationships between the families Eulophidae,
Tetracampidae and Trichogrammatidae, but at least Eulophidae and
Trichogrammatidae are supported as monophyletic. Also, the Eulophidae-lineage
appeared to consist of four internal lineages corresponding to
the recognized subfamilies: Eulophinae, Tetrastichinae, Entedoninae
and Entiinae (= Euderinae), what was also supported by morphological
characters (including putative synapomorphies). Largely similar
pattern was reported by other students foe Eulophidae (Burks et
al., 2011) and Chalcidoidea as a whole (Munro et al., 2011; Heraty
et al., 2013).
Our
research (Gumovsky, 2011) based on the combined analysis of nuclear
(28S D2 rDNA) and two mitochondrial (cytochrome oxidase subunit
I, COI, and cytochrome b, Cyt b) sequences demonstrated that there
is no signal of close relationships between the families Eulophidae,
Tetracampidae and Trichogrammatidae, but at least Eulophidae and
Trichogrammatidae are supported as monophyletic. Also, the Eulophidae-lineage
appeared to consist of four internal lineages corresponding to
the recognized subfamilies: Eulophinae, Tetrastichinae, Entedoninae
and Entiinae (= Euderinae), what was also supported by morphological
characters (including putative synapomorphies). Largely similar
pattern was reported by other students foe Eulophidae (Burks et
al., 2011) and Chalcidoidea as a whole (Munro et al., 2011; Heraty
et al., 2013).